- Your cart is empty
- Continue Shopping
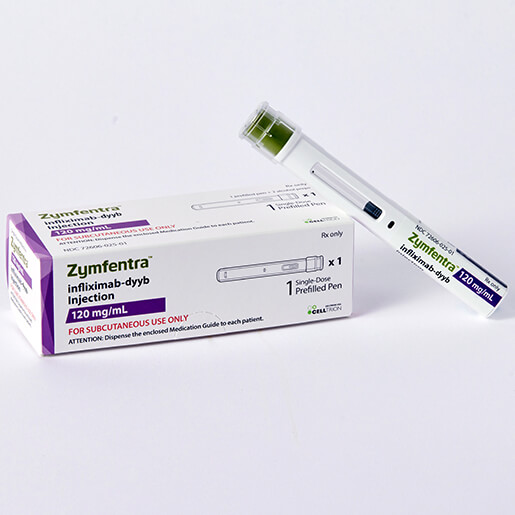
Zymfentra
$800.00Current price is: $800.00. Original price was: $835.00.
Zymfentra™ (infliximab-dyyb) is the first and only FDA-approved subcutaneous infliximab biosimilar.
After two starter IV infusions, patients can self-inject Zymfentra every 2 weeks at home – delivering the same proven efficacy as Remicade® with greater convenience for adults with moderately to severely active Crohn’s disease or ulcerative colitis.
One 120 mg/mL prefilled syringe or autoinjector. No infusion center visits required after induction.
Product Description
Zymfentra (vedolizumab-mttf) represents a significant advancement in the management of inflammatory bowel disease (IBD). It is the first FDA-approved subcutaneous formulation of vedolizumab, specifically indicated as a maintenance therapy for adults with moderately to severely active Ulcerative Colitis (UC) or Crohn’s Disease (CD).
The primary benefit of Zymfentra is flexibility. Unlike traditional maintenance therapies that require regular visits to an infusion center, Zymfentra is designed for self-administration. After completing an initial induction phase with intravenous vedolizumab to bring symptoms under control, patients can transition to Zymfentra for their maintenance dosing. Available in a single-dose prefilled pen or prefilled syringe, it allows patients to manage their condition from the comfort of their own home, providing effective long-term care with fewer interruptions to daily life.
Key benefits:
Same trusted TNF-blocker mechanism as Remicade®
Proven remission rates in Crohn’s disease and ulcerative colitis
Self-injection at home after initial IV loading
No new safety signals relative to reference infliximab
Available in refrigerated 120 mg/mL single-dose format
Zymfentra represents a new era of flexibility for IBD patients who have responded to IV infliximab. Speak to your gastroenterologist today to see if switching to Zymfentra subcutaneous maintenance therapy is right for you.








Reviews
There are no reviews yet.